Figure 1 from Hydrolysis and transesterification of parabens in an aqueous solution in the presence of glycerol and boric acid | Semantic Scholar

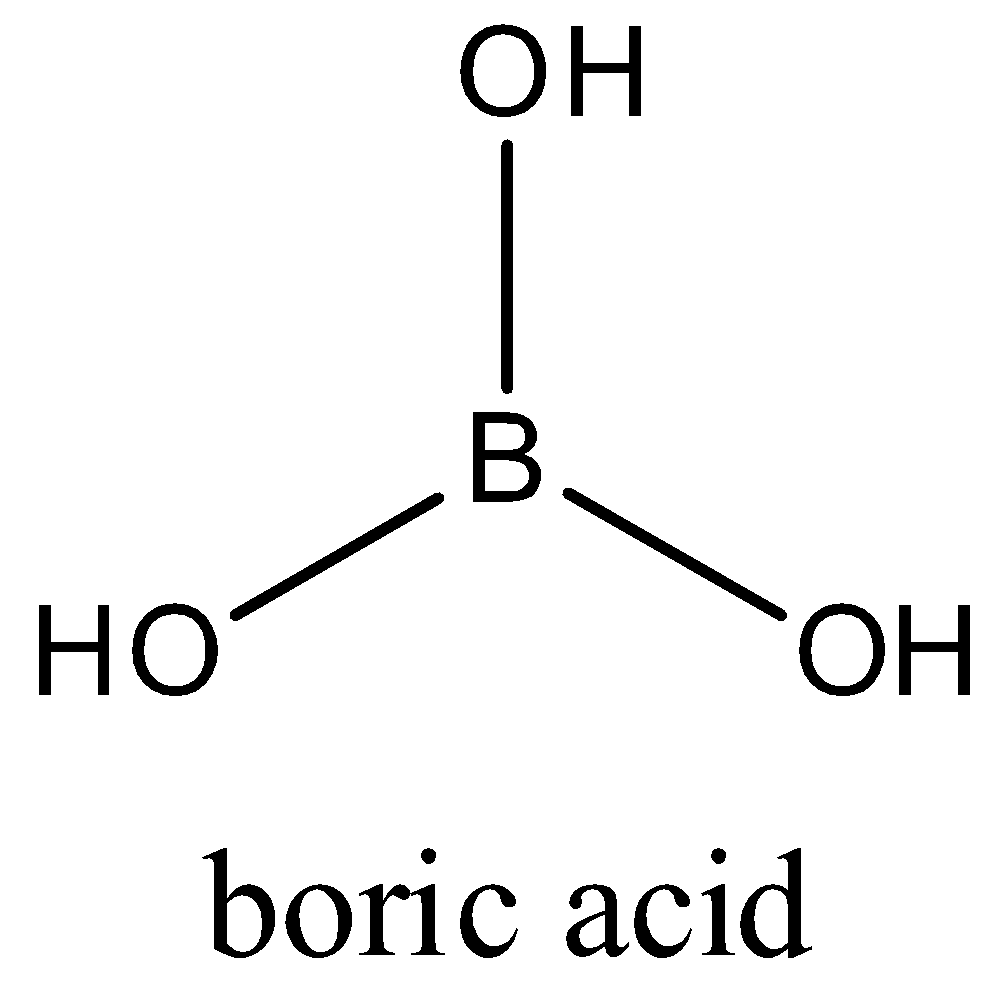
Boric Acid, a Lewis Acid With Unique and Unusual Properties: Formulation Implications - Journal of Pharmaceutical Sciences

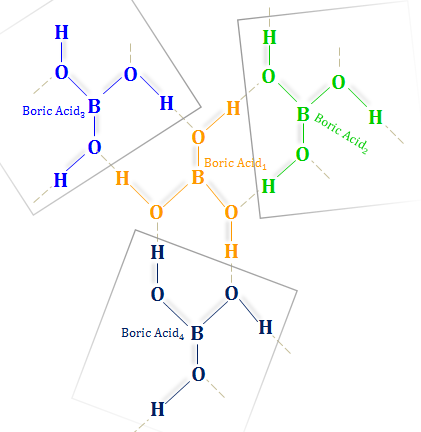
Novel (Glycerol)borate-Based Ionic Liquids: An Experimental and Theoretical Study | The Journal of Physical Chemistry B
Incorporation of stable glycerol-borate complex into phenolic resin as a glue line treatment in engineered wood products

Novel (Glycerol)borate-Based Ionic Liquids: An Experimental and Theoretical Study | The Journal of Physical Chemistry B
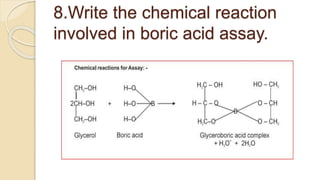
Hydrolysis and transesterification of parabens in a aqueous solution in the presence of glycerol and boric acid | Journal of Excipients and Food Chemicals

what happens if boric acid is added to glycerol explain reaction involved explain meachanism for rxn - Chemistry - Hydrocarbons - 10848765 | Meritnation.com

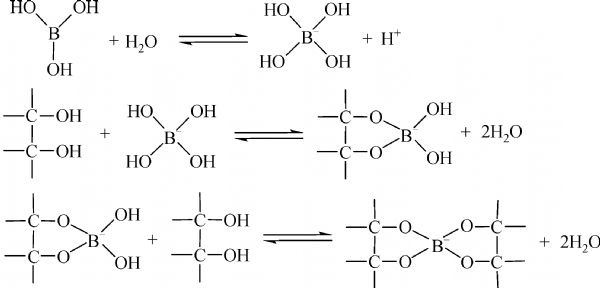
Release of the potential acidity of (weak) boric acid via exaltation... | Download Scientific Diagram

Effect of ultra-violet cross-linking on the properties of boric acid and glycerol co-plasticized thermoplastic starch films - ScienceDirect

Boric acid can be titrated against sodium hydroxide using phenolphthalein as an indicator only in the presence of polyhydroxy compounds like glycol. Glycerol etc. Give reason.

Highly efficient one-pot, three-component Mannich reaction catalysed by boric acid and glycerol in water with major 'syn' diastereoselectivity - ScienceDirect

42220-19-9 Boric acid (H3BO3), reaction products with glycerol C3H11BO6, Formula,NMR,Boiling Point,Density,Flash Point

Release of the potential acidity of (weak) boric acid via exaltation... | Download Scientific Diagram
Welcome to Chem Zipper.com......: Why Boric acid become strong acid in the presence of cis 1,2-diol or 1,3-diol ?
Boric Acid / Glycerol as an Efficient Catalyst for Synthesis of Thiomorpholine 1,1-Dioxide by Double Michael Addition Reaction i

PDF) Boric acid/glycerol as an efficient catalyst for regioselective epoxide ring opening by aromatic amines in water
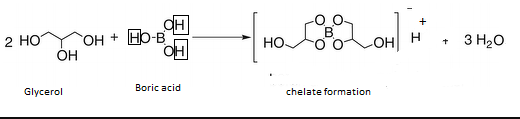
Boric acid is a very weak acid but in the presence of certain organic compounds, it acts as a strong acid. Which one of the following organic compound(s) affect cannot such (A)